What is luminescence?
In modern physics light is assigned two qualities: A wave quality and a particle quality. The particle quality means that light consists of certain energy packages, so-called light quanta or photons. The energy that is contained in these photons is reflected in their wavelength – the physical distance between wave trough and wave crest. A short wavelength means a high energy content, a large wavelength means a low energy content.
- The photon passes through the matter without hindrance and in the same direction. (Transmission, example: Light impinges on glass)
- The photon hits an atom and is reflected by this atom with the same energy in a different direction. (Reflection, example: Light impinges on a mirror)
- The photon is swallowed by the matter. The energy of the photon in this case is transferred to the matter and leads to an increase of the matter's energy content, either in the form of heating and/or electric excitation of the matter.
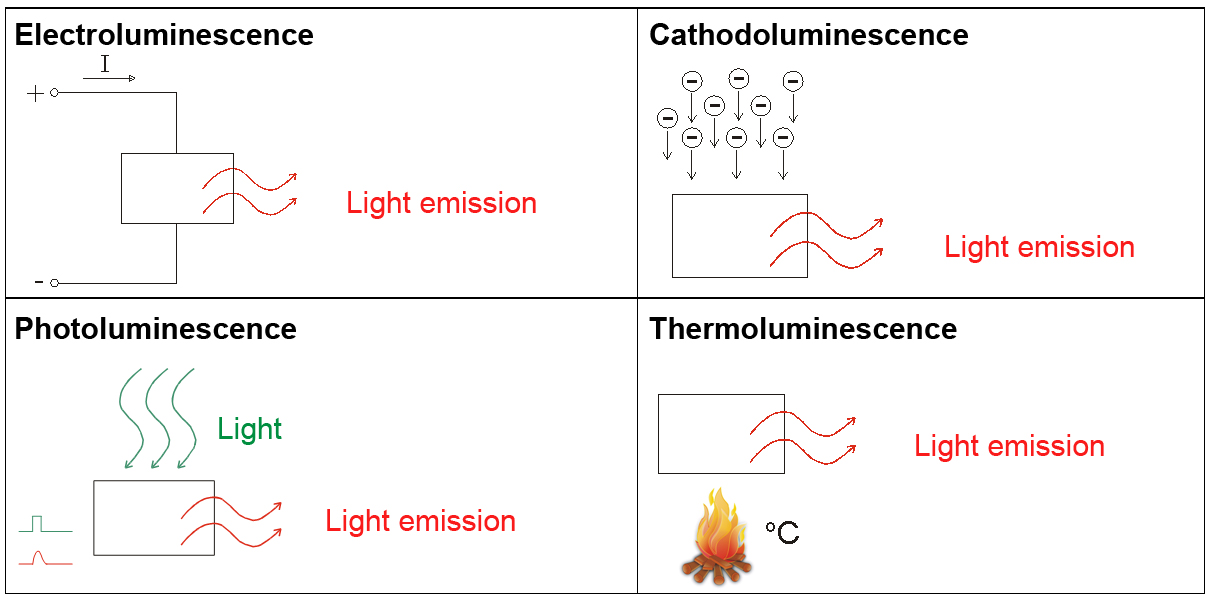
- Electric current (electroluminescence) e.g. in LEDs: Transition from valence band to conduction band
- Bombardment with electrons (cathodoluminescence) e.g. cathode ray tube
- Light, i.e. bombardment with photons (photoluminescence)
Fluorescence means that the emission of photons by the excited material stops immediately or within a few nanoseconds after excitation ends.
Phosphorescence means that the emission of light continues beyond the nanosecond range and extends into the millisecond or even the second range.
Addition of heat (thermoluminescence)
When electromagnetic radiation (light from ultraviolet to infrared) impinges on an object, part of the light is absorbed. The major part of the absorbed light is converted into heat (a black surface absorbs more light than for example a white surface and thus becomes correspondingly warmer when it is irradiated with light).
The light essentially is absorbed by molecules. This additional energy excites the molecules to move faster, i.e. they oscillate at a higher rate, which involves an increase of temperature.
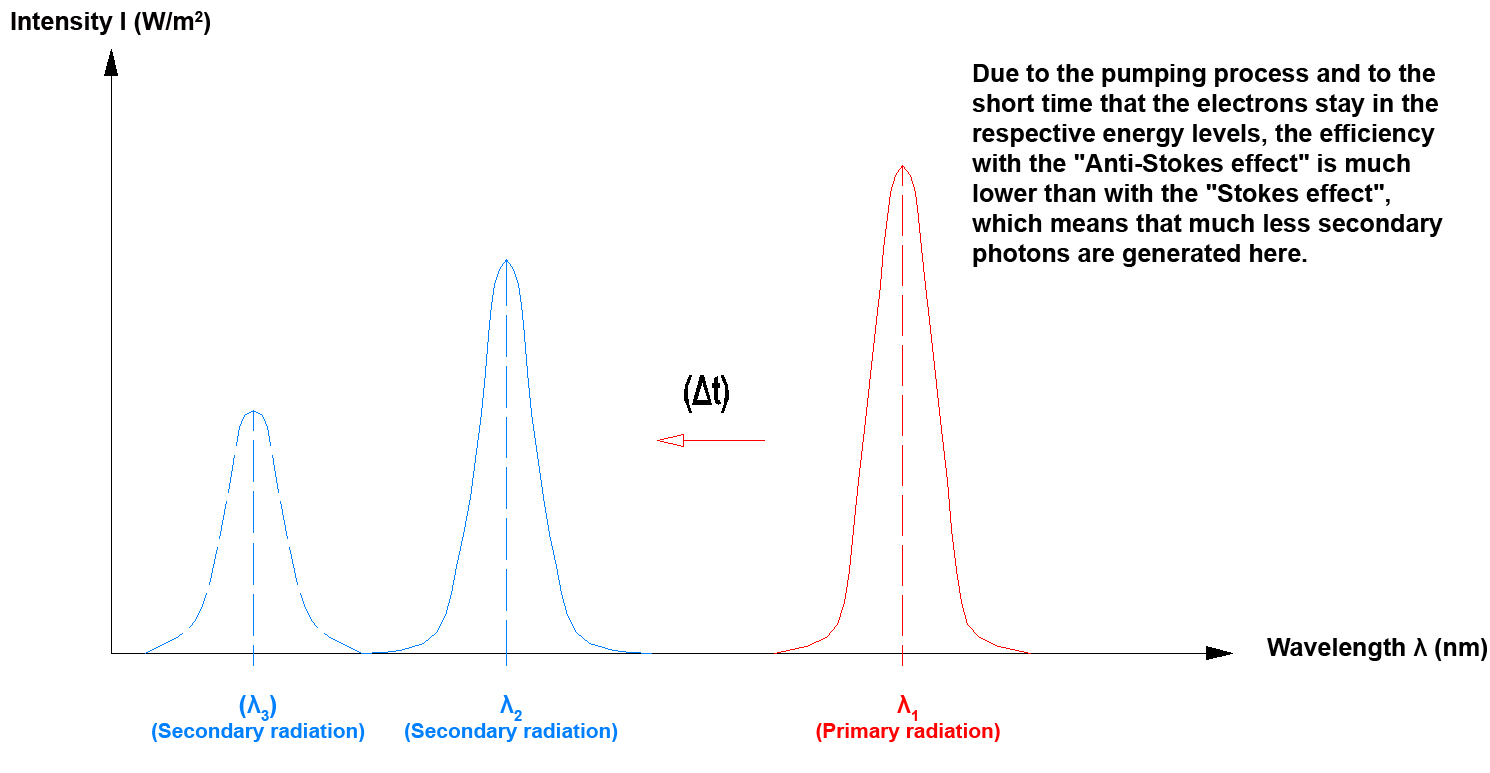
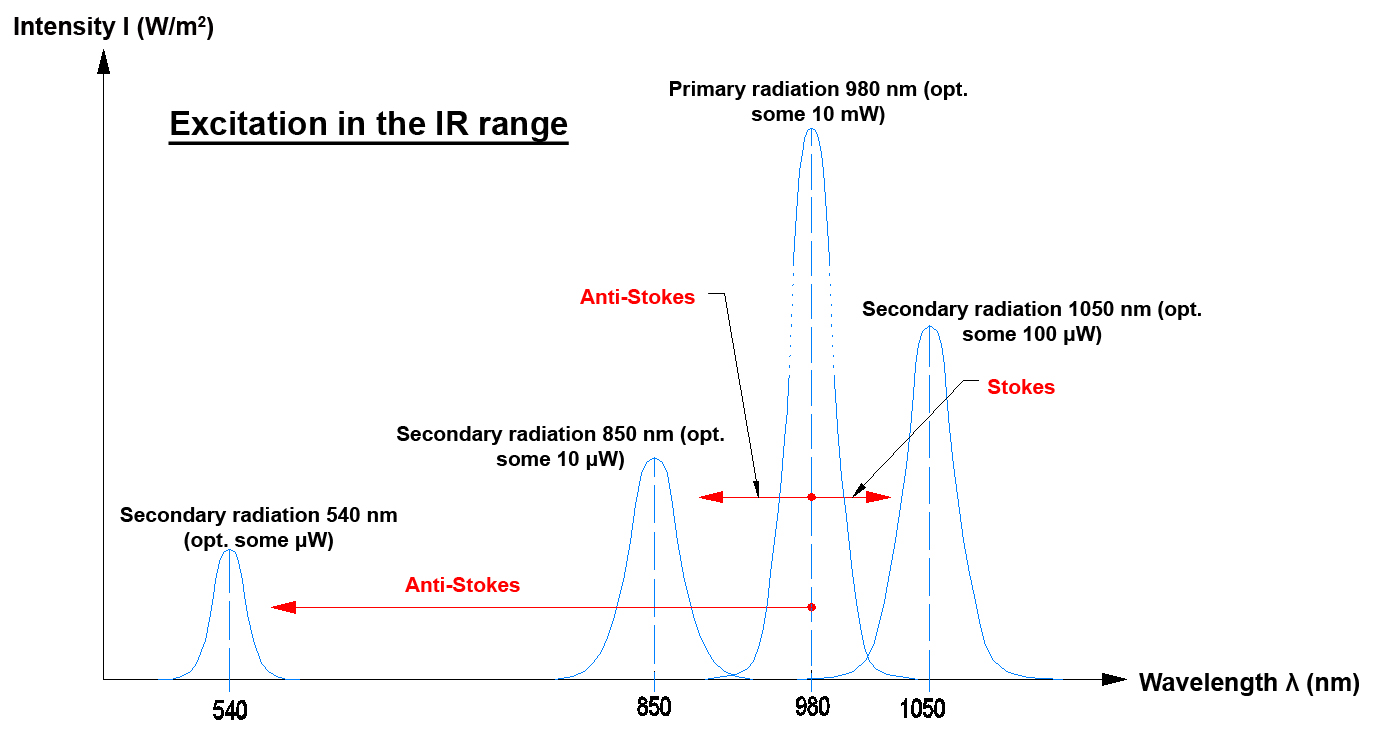
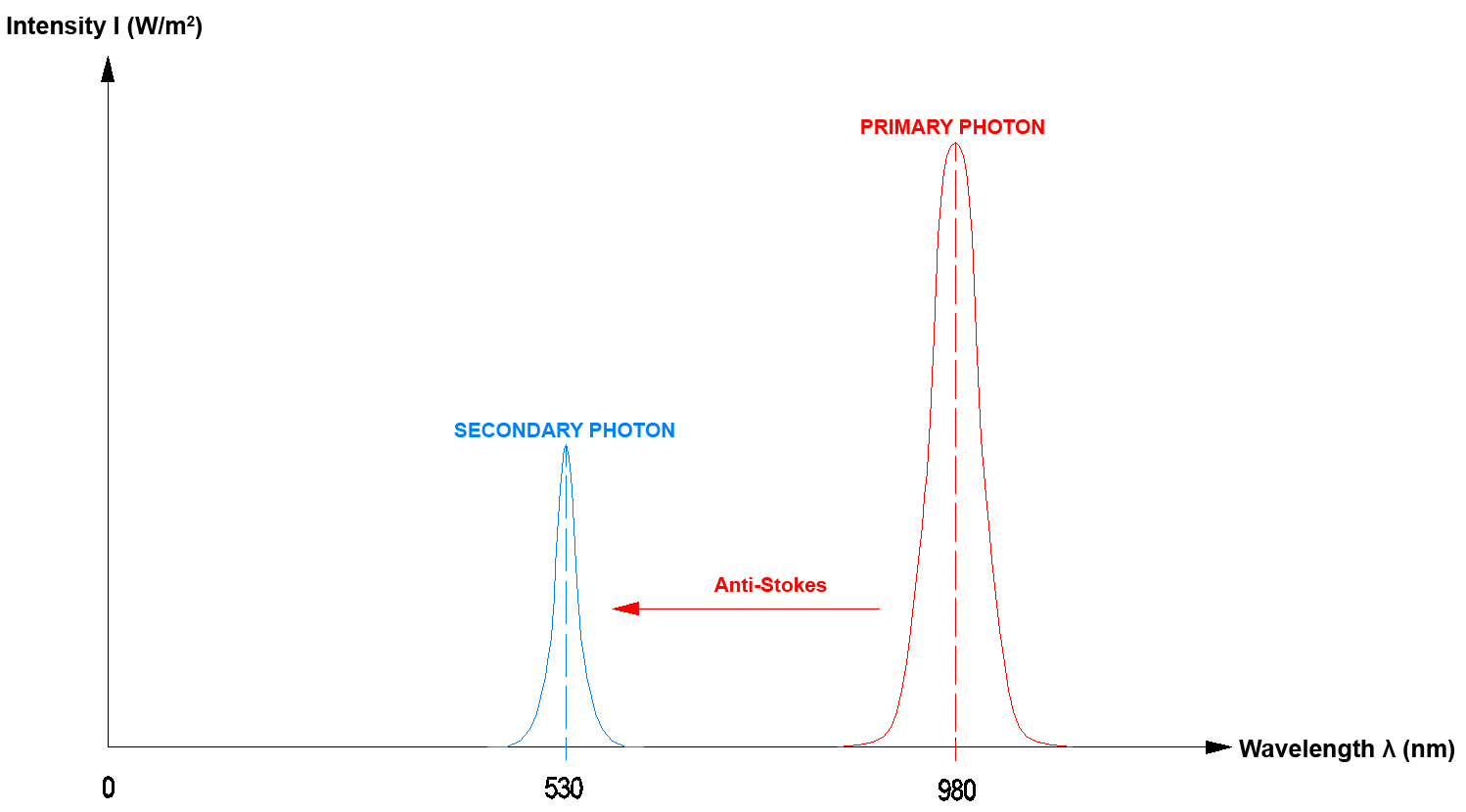
If electromagnetic radiation that impinges on an object and is absorbed by this object has a longer wavelength than the electromagnetic radiation that is emitted by the object, this is referred to as an "Anti-Stokes effect".
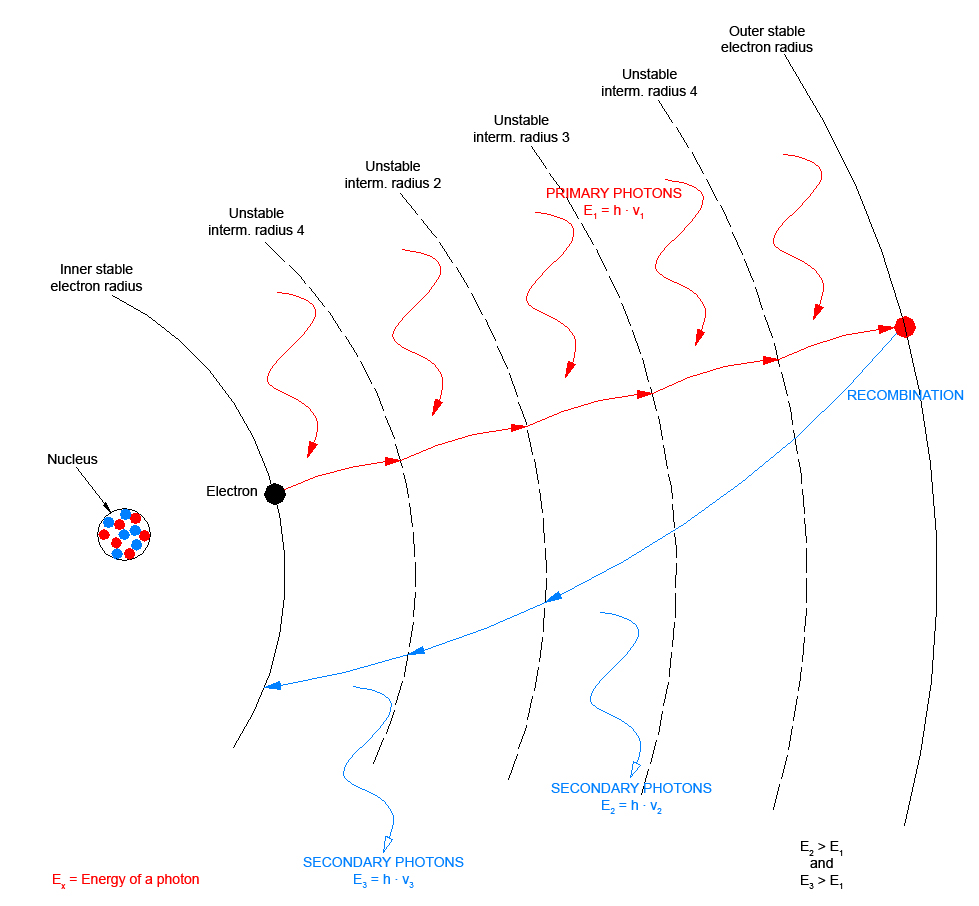
these radiuses only for a relatively short time before they return to their initial state again.
This so-called recombination typically lies in the nano-, micro- or millisecond range.
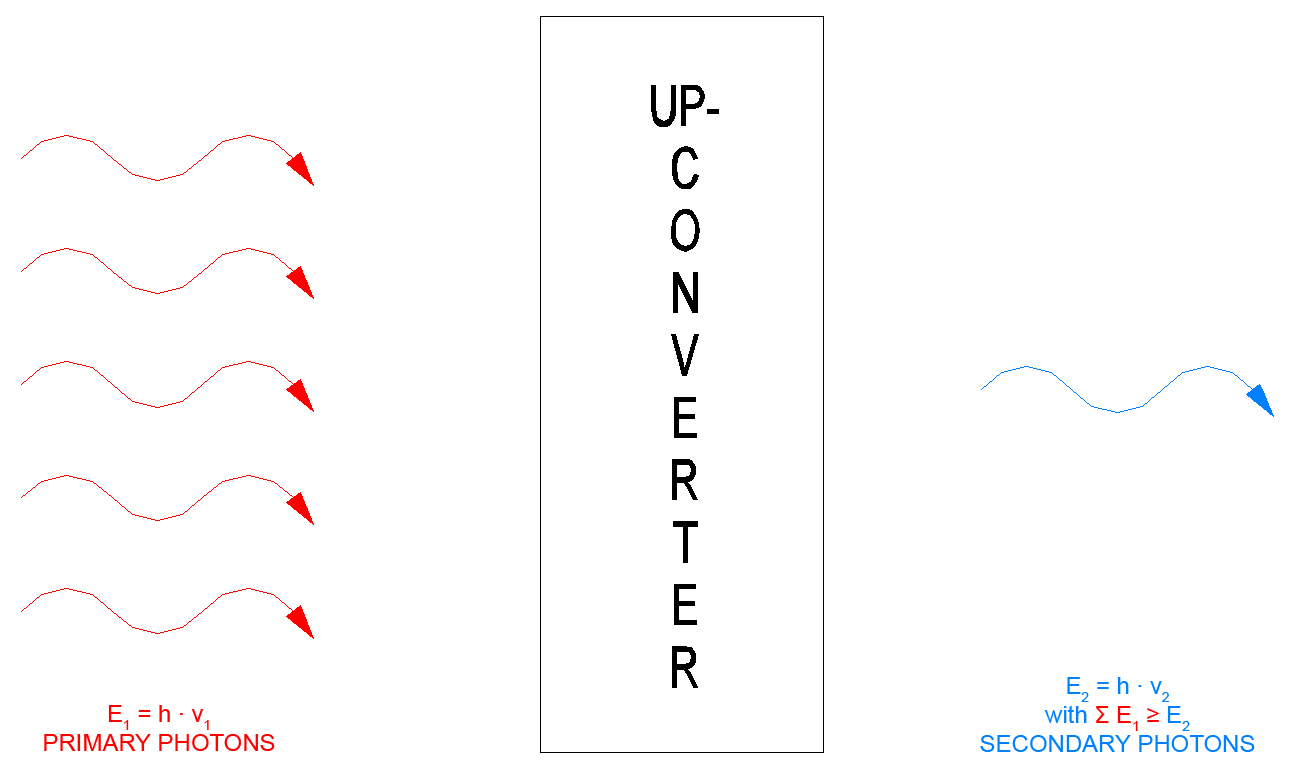
Since with the "Anti-Stokes effect" several photons are required to move an electron to an outer electron radius, this also is referred to as a so-called pumping process.
During "pumping" the primary photons repeatedly lift the electrons from one intermediate radius to the next, they so to speak pump the electrons to the outer electron radius. The further outside the electron path lies, the higher is the potential energy of the electron on the path. It has a higher energy level. A higher difference between two energy levels means a higher energy of the photon that is generated when the electron returns to its stable level.
What are "rare earth metals"?
Rare earth metals are chemical elements of the 3rd group in the periodic table; lanthanides
(17 elements) also belong to the rare earths.
Among others (rare earths for example also are needed for permanent magnets) the rare earth metals also have special spectroscopic properties. Unlike semiconductors (the band gap, i.e. the energy difference between valence band and conduction band of a semiconductor depends on the temperature, and thus also the wavelength that is emitted by the semiconductor crystal) rare earths have a discrete energy spectrum (i.e. defined energy levels in the electron shell) in the solid body (crystal). This means that for the absorption of electromagnetic radiation a certain wavelength is required that depends on the difference of the energy levels. Optical energy emission (to the outside) also is performed in discrete wavelengths that are determined by the crystal.
| Y 39 | Yttrium |
(among others used for luminescent materials and LEDs) |
| Pm 61 | Promethium | (used for luminous dials) |
| Eu 63 | Europium | (used in LEDs and luminescent materials) |
| Gd 64 | Gadolinium | (used for green luminescent materials) |
| TB 65 | Terbium | (used for luminescent materials) |
| Tm 69 | Thulium | (luminescent materials for TV sets) |
| Yb 70 | Ytterbium | (luminescent materials for TV sets) |
The optical properties can be correspondingly influenced and specifically changed by targeted doping of the rare earth crystals. Erbium, for example, is used as a so-called activator. This activator so to speak acts as a "catalyst".
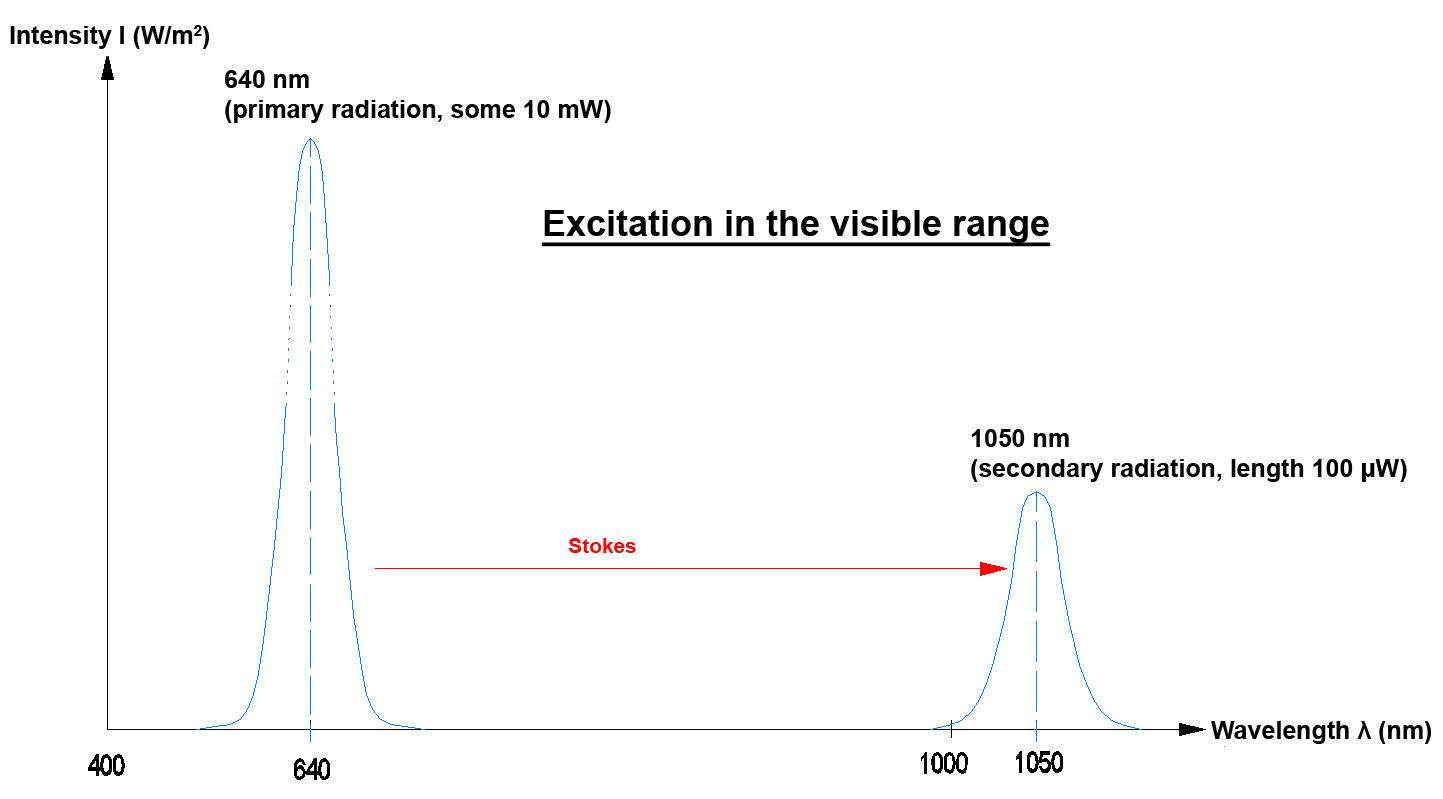
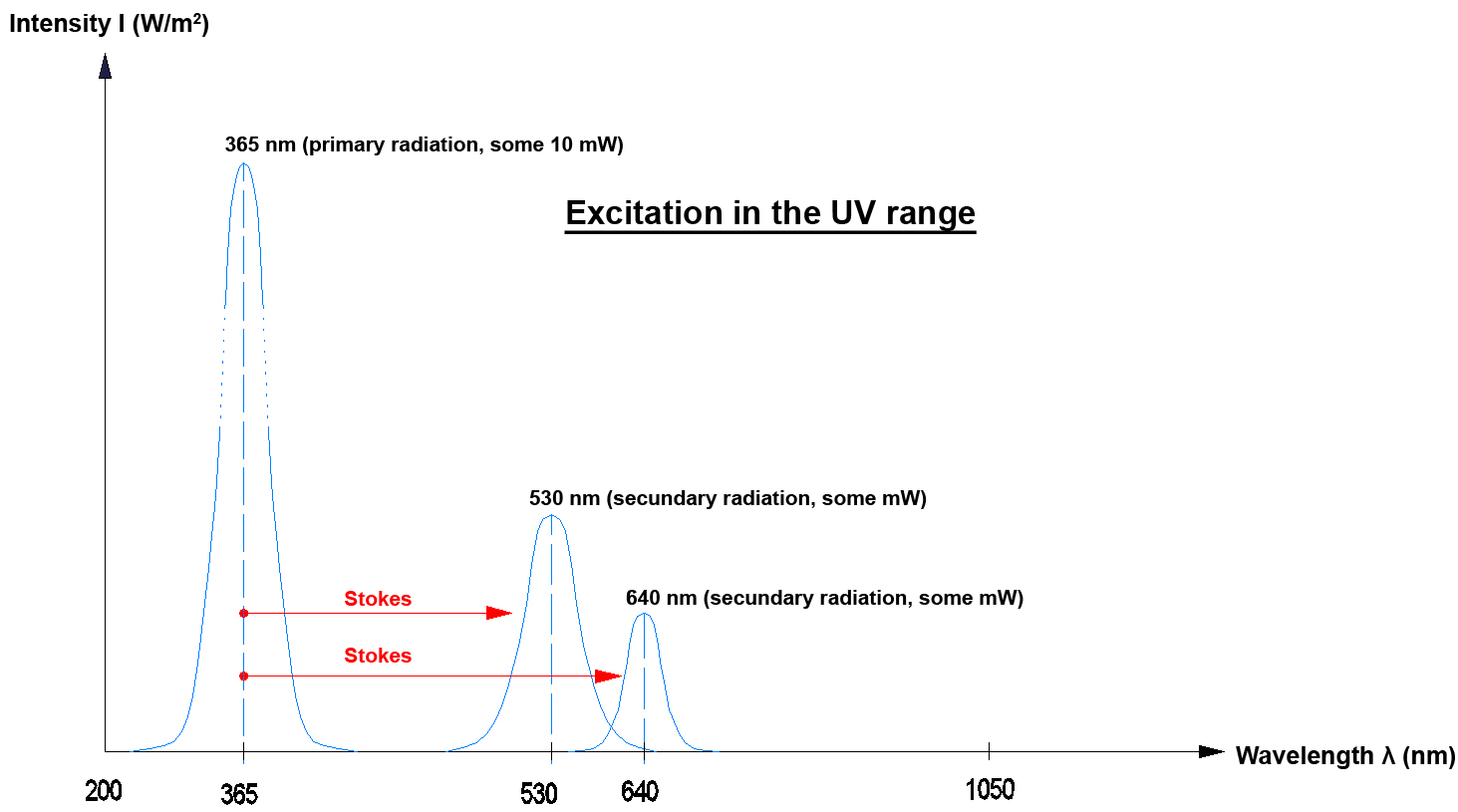
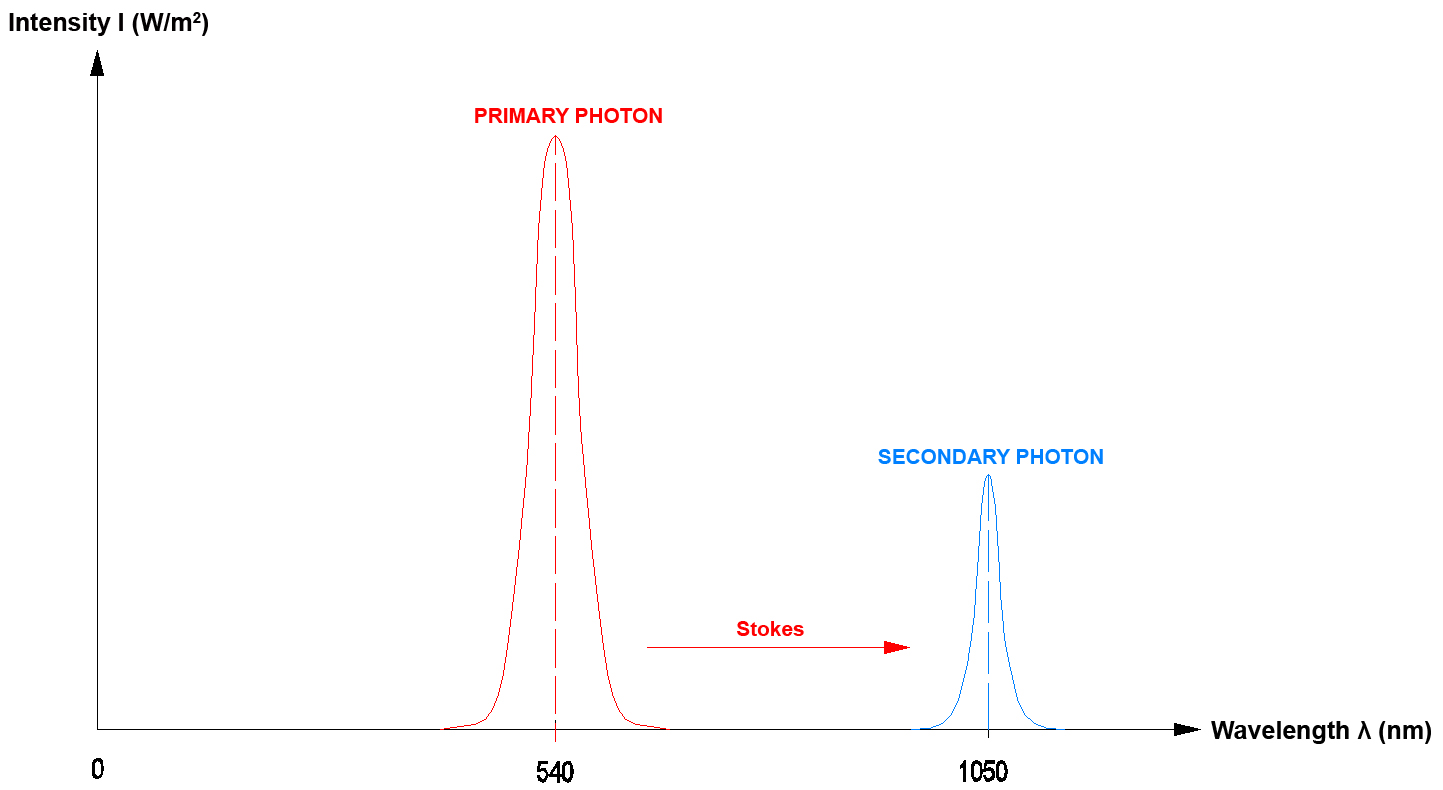
In practice the "Stokes effect" and the "Anti-Stokes effect" frequently occur when photons are emitted.
Usually there is a difference of some decades between the optical power that is required for exciting the process and the optical power that is actually emitted by the crystal. The optical power required for excitation in the IR range approximately is 10 mW (e.g. with 980 nm), the emission then lies in the visible wavelength range with some µW (Anti-Stokes). In the IR range, however, the emission also may come close to the mW range (Stokes). A part of the primary radiation first also is used to excite the crystal, photons are thus used to generate phonons, which increases the lattice oscillation (crystal). In the following step photons (light) may in turn be generated by phonons (lattice oscillation).
In an optical sense an "UP-Converter" converts light (from the UV to the IR range) of a longer wavelength (i.e. with lower-energy photons) into a shorter wavelength (the photons then have more energy). This energy gain is achieved by way of "optical pumping". In order to emit a secondary photon the so-called up-converter must absorb the number of primary photons that is necessary for reaching the energy level of the secondary photon (until the electron has been lifted to the outer electron shell). This also is referred to as the so-called Anti-Stokes effect.
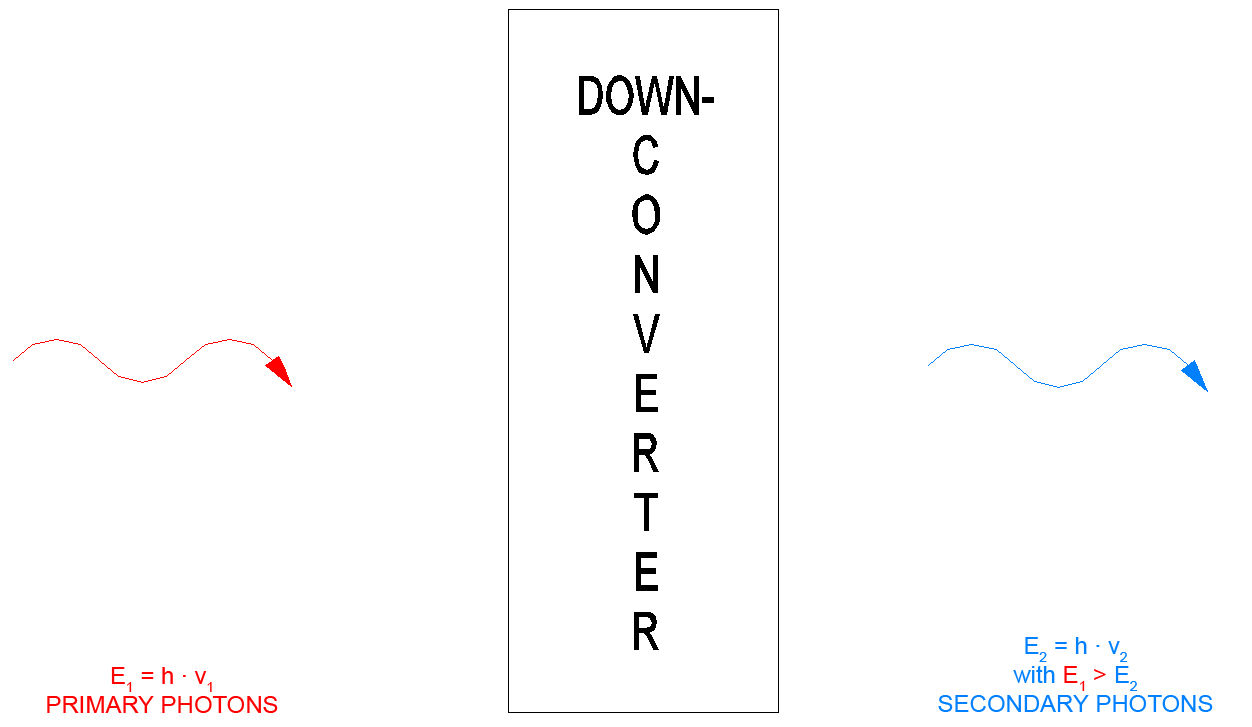
In optics a "DOWN-Converter" means that the secondary photon that is generated by a primary photon has less energy. This means that only one primary photon is necessary to generate a secondary photon. The energy difference for example is given off into the lattice of the DOWN-Converter crystal (as a phonon increase of lattice oscillation). The process in the DOWN-Converter can be explained with the Stokes effect.
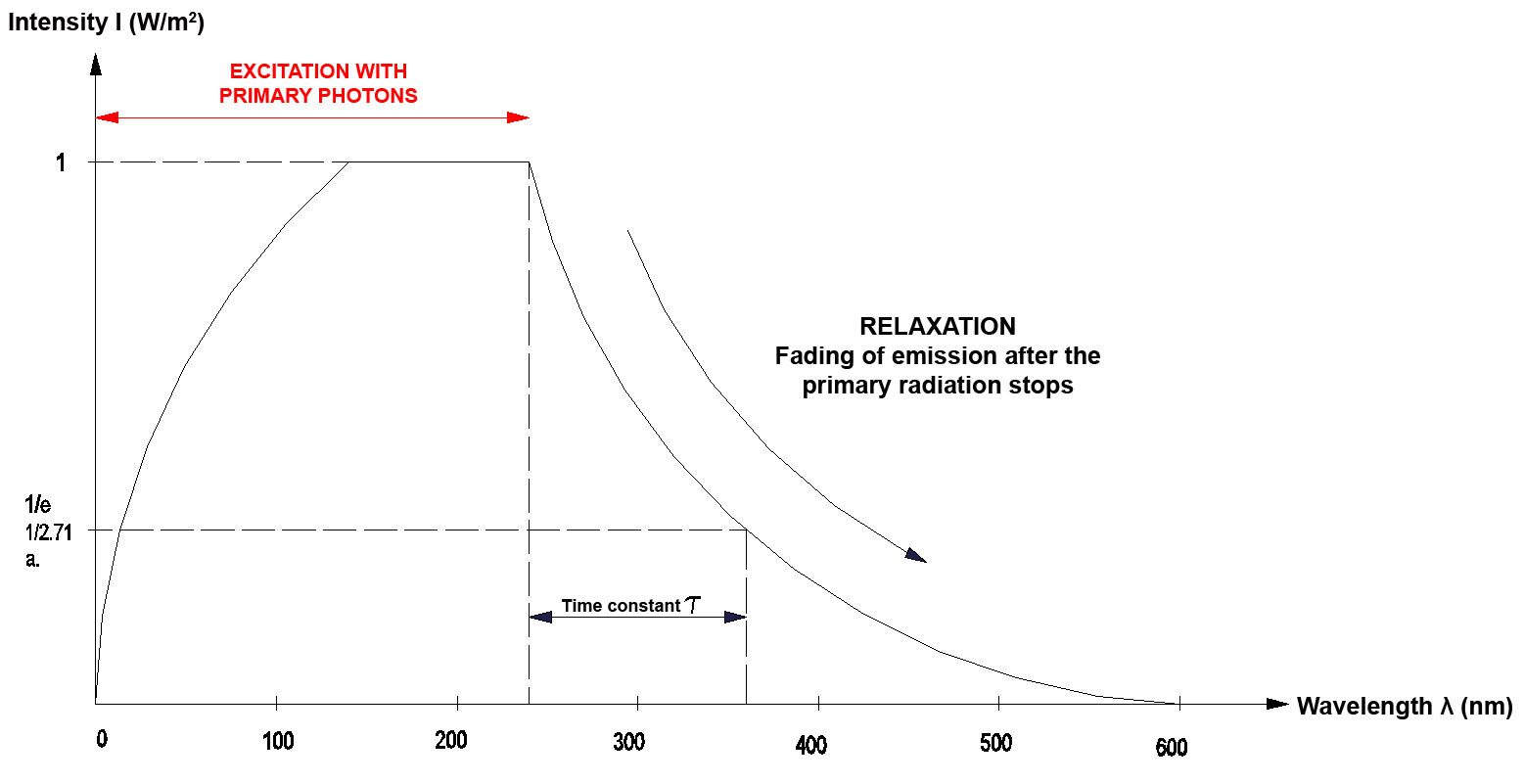
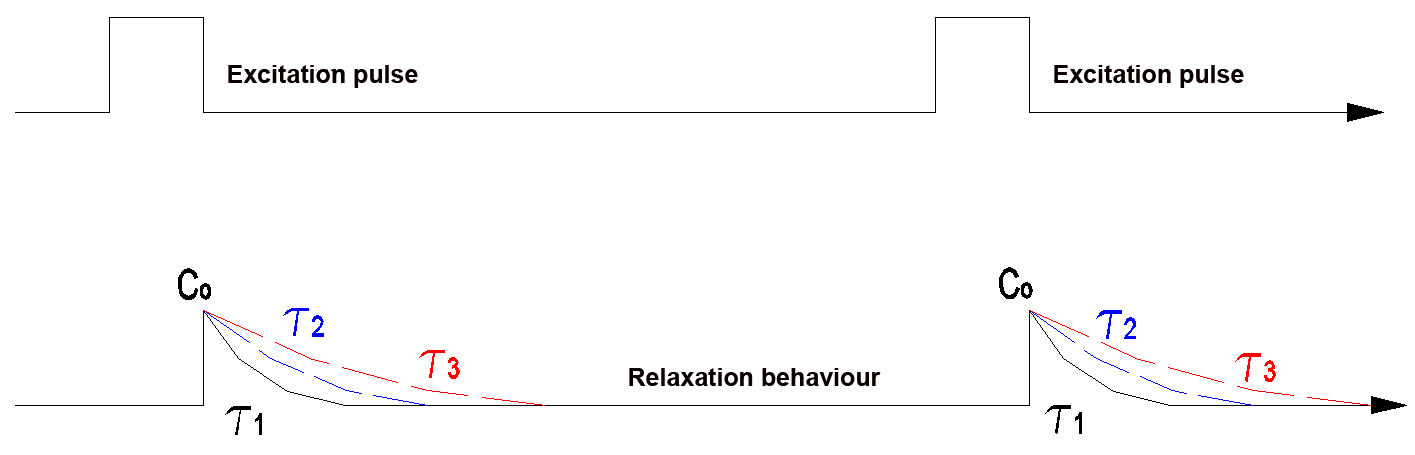
In optics the relaxation time is the time that an object needs to return from the excited state to the initial state again. The time constant t is a measure for this time. It describes how long it takes until a process - starting from the excited state - has faded to approximately a factor of 1/2.71 or to 1/e (e is the so-called Euler number). The time constant usually is used for processes that fade exponentially. Practice has shown that excitation processes that follow both the Stokes and the Anti-Stokes effect approximately fade exponentially.
It should never be forgotten, however, that the primary radiation that is used for this purpose
lies in the laser class 3B range. The laser pen is a product of laser class 3B.
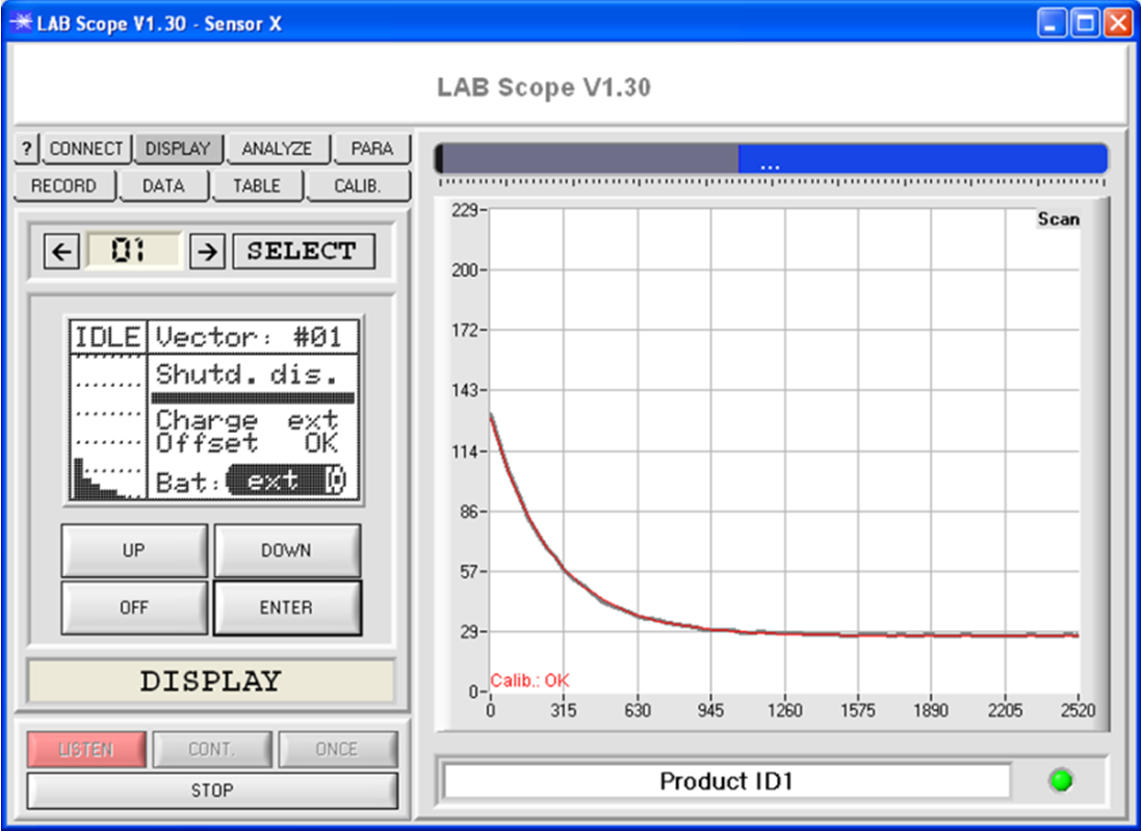
A special monitoring software can be used to record measurement data.
Both detection devices belong to laser class 1, which is achieved by means of a special, patented process.
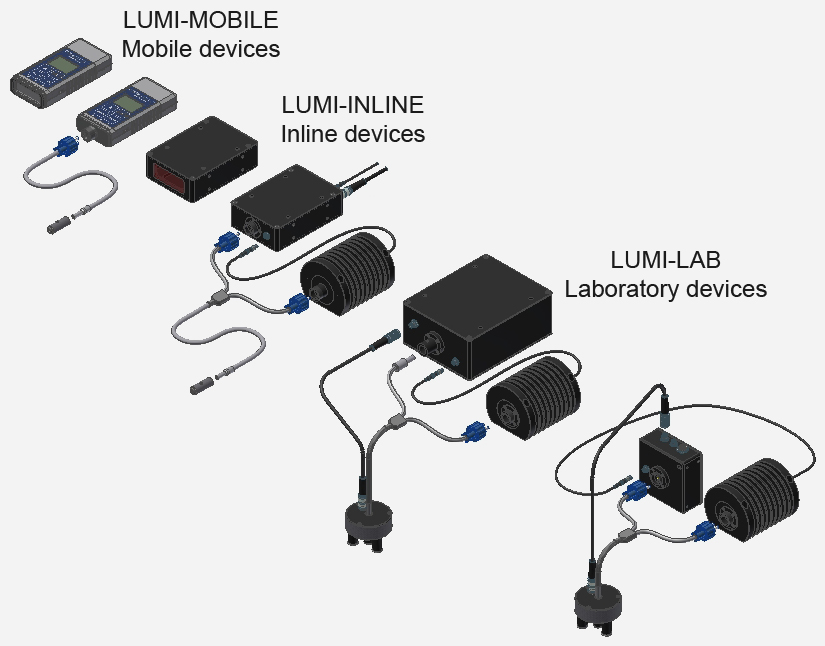
Detection devices for mobile use
An IR pulse or UV pulse is directed onto the object to be inspected. After the end of the excitation pulse the relaxation curve is recorded and evaluated. The initial intensity of the relaxation pulse also is determined.

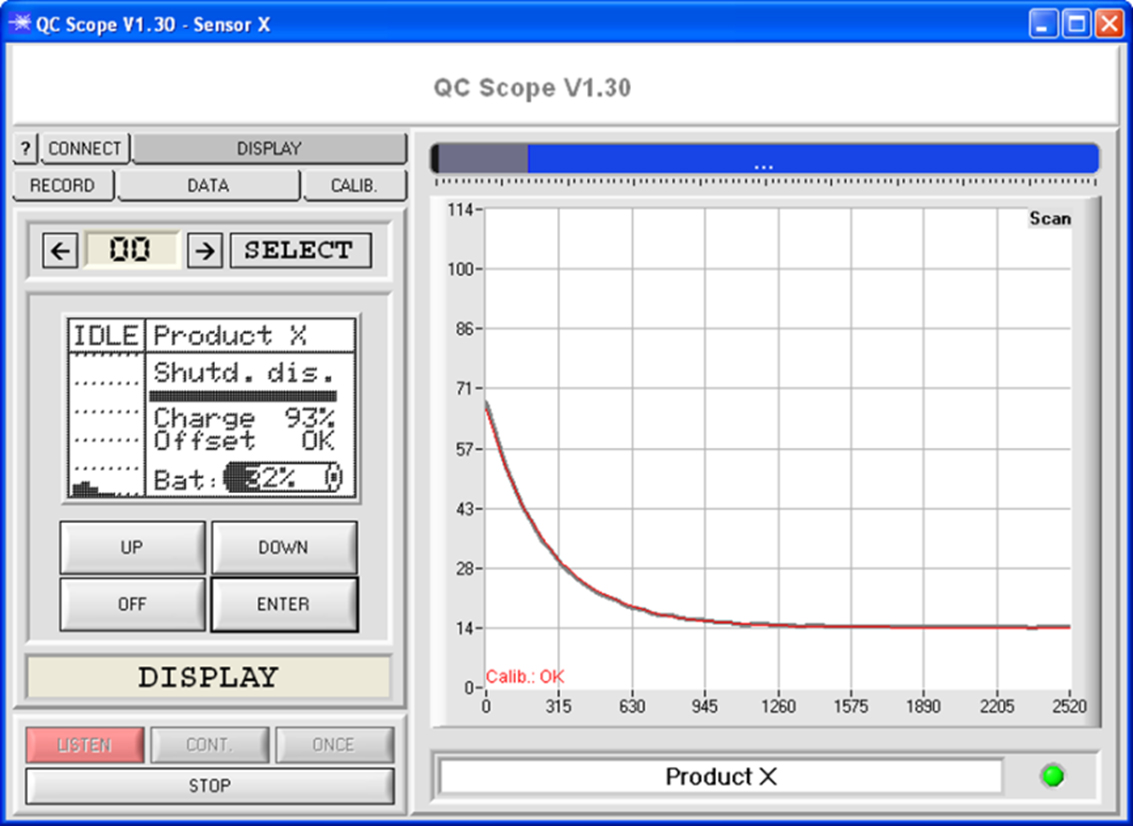
These devices can save up to 31 products that can be read from a file (the products have been determined with a LUMI-MOBILE-LAB device and saved in a file before).
The detection of a product is indicated both acoustically and optically on the graphic display. The devices check the relaxation behaviour of the rare earth crystal.
LUMI-MOBILE-PT-UV/VIS

Unlike the LUMI-MOBILE-QC devices these detection devices work with one parameter set.
LUMI-MOBILE-DA-UV/VIS

Unlike the LUMI-MOBILE-QC and LUMI-MOBILE-PT devices these detection devices do not search for certain products that have first been determined by a LUMI-MOBILE-LAB device. These devices generally search for a time constant and for a concentration of a rare earth crystal, and they do this over the complete dynamic range, i.e. from a very low concentration in the ppm range up to a very high concentration.
Windows® software LUMI-MOBILE-DA-Scope
LUMI-MOBILE-JR-UV/VIS

Windows® software LUMI-MOBILE-JR-Scope
LUMI-MOBILE-FIO-LAB-UV/VIS

The LUMI-MOBILE-FIO-LAB optical-fibre device essentially is identical to the LUMI-MOBILE-LAB standard device. Only the optics have been modified for optical-fibre connection.
LUMI-MOBILE-FIO-QC-UV/VIS

The LUMI-MOBILE-FIO-QC optical-fibre version has the same software as the LUMI-MOBILE-QC, only the hardware in this device features an optical-fibre connection.
LUMI-MOBILE-FIO-PT-UV/VIS

The LUMI-MOBILE-PT also is available as an optical-fibre version LUMI-MOBILE-FIO-PT.
The software is the same as in the LUMI-MOBILE-PT standard version.
LUMI-MOBILE-FIO-DA-IR/IR
LUMI-MOBILE-FIO-DA-UV/VIS

The LUMI-MOBILE-PT also is available as an optical-fibre version LUMI-MOBILE-FIO-PT.
The software is the same as in the LUMI-MOBILE-PT standard version.
LUMI-MOBILE-FIO-JR-UV/VIS

The LUMI-MOBILE-JR also is available as an optical-fibre version LUMI-MOBILE-FIO-JR.
The optical-fibre version has the same software as the LUMI-MOBILE-JR standard version.
INLINE detection devices for production lines
LUMI-INLINE-IR/IR
LUMI-INLINE-UV/VIS


The LUMI-INLINE versionis equipped with two analog outputs that provides information about the concentration of the rare earth crystal, and with 4 digital outputs that can be used to save up to 15 products.
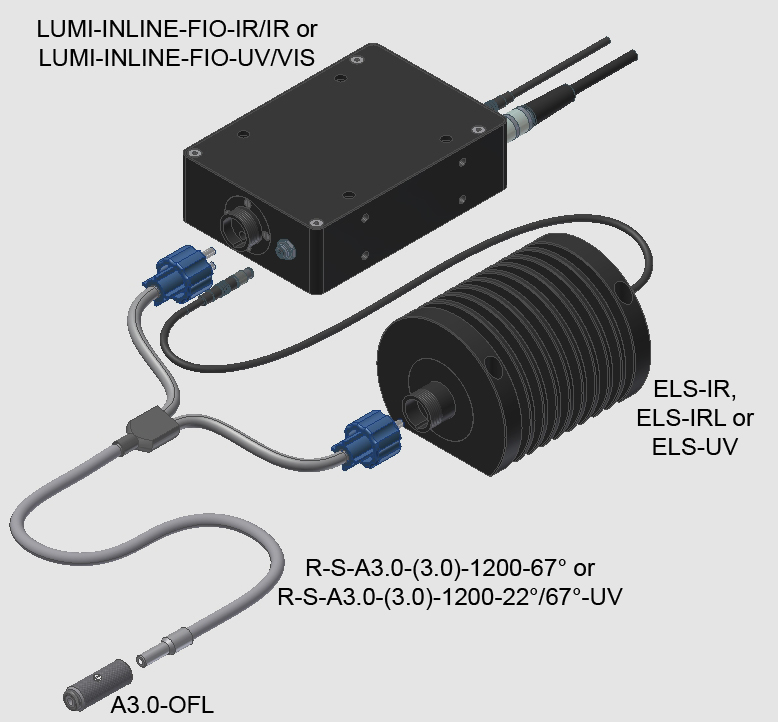
LUMI-INLINE-FIO-IR/IR
LUMI-INLINE-FIO-UV/VIS
Windows® software LUMI-INLINE-Scope
Detection devices for the laboratory
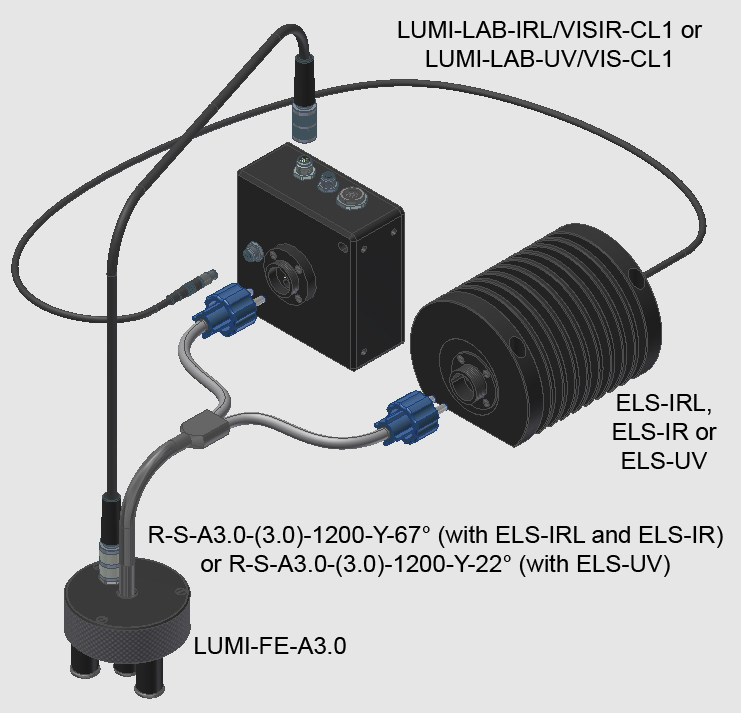
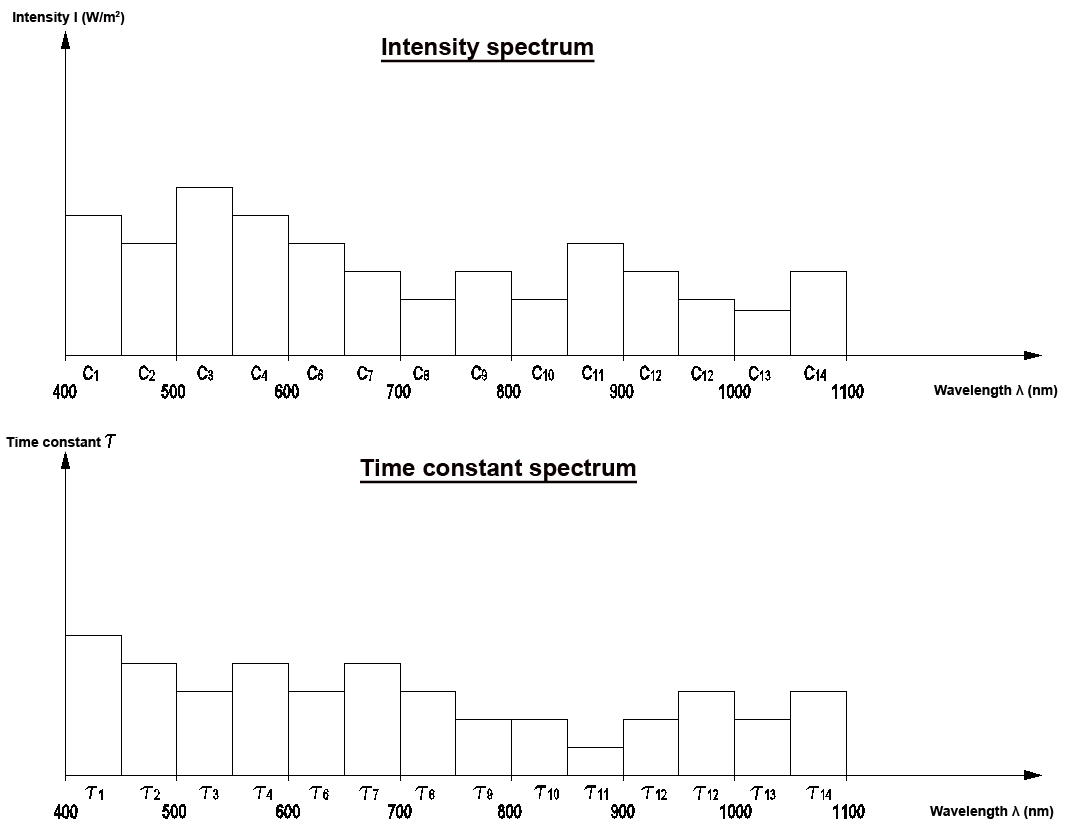
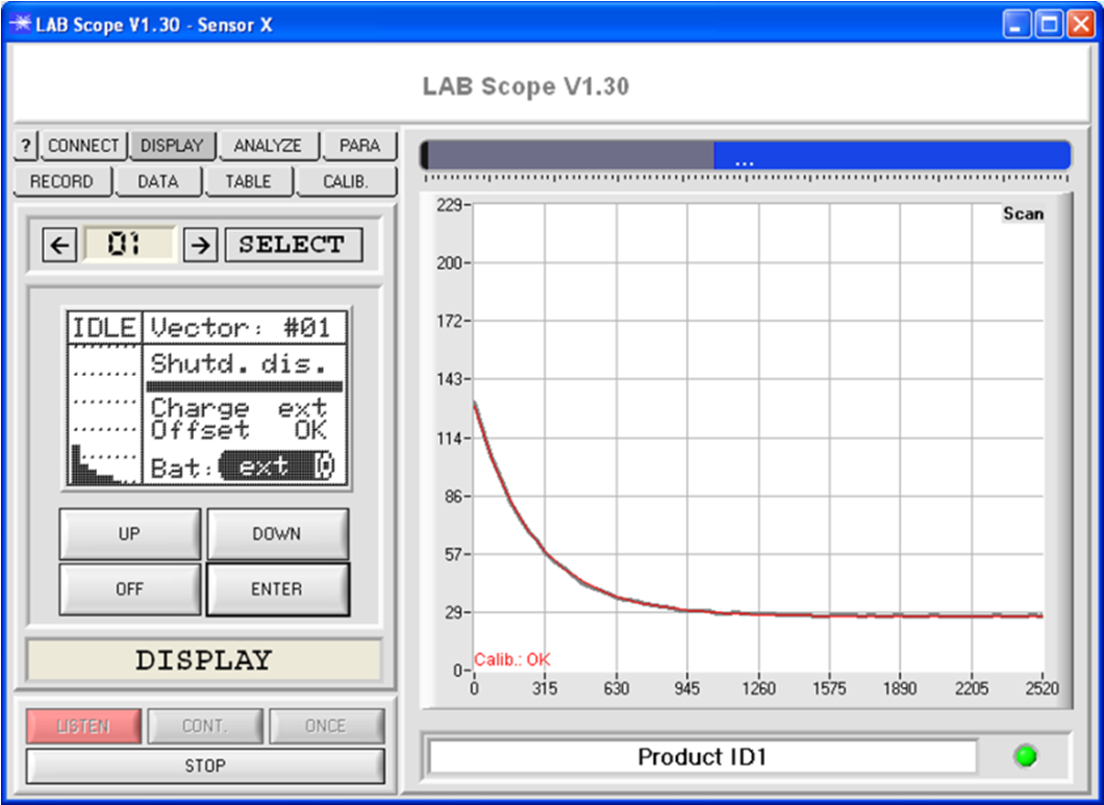
These LUMI-LAB laboratory devices allow the representation of the secondary spectrum in the visible range. The LUMI-LAB-IRL/VISIR furthermore records the relaxation times in the IR range, and the LUMI-LAB-UV/VIS represents the relaxation curves in the visible spectrum range.
LUMI-LAB-IRL/VISIR-CL1
LUMI-LAB-UV/VIS-CL1
Corresponding software:
Windows® software LUMI-LAB-Scope
LUMI-LAB-8-IRL/IR-CL1
LUMI-LAB-8-IR/IR
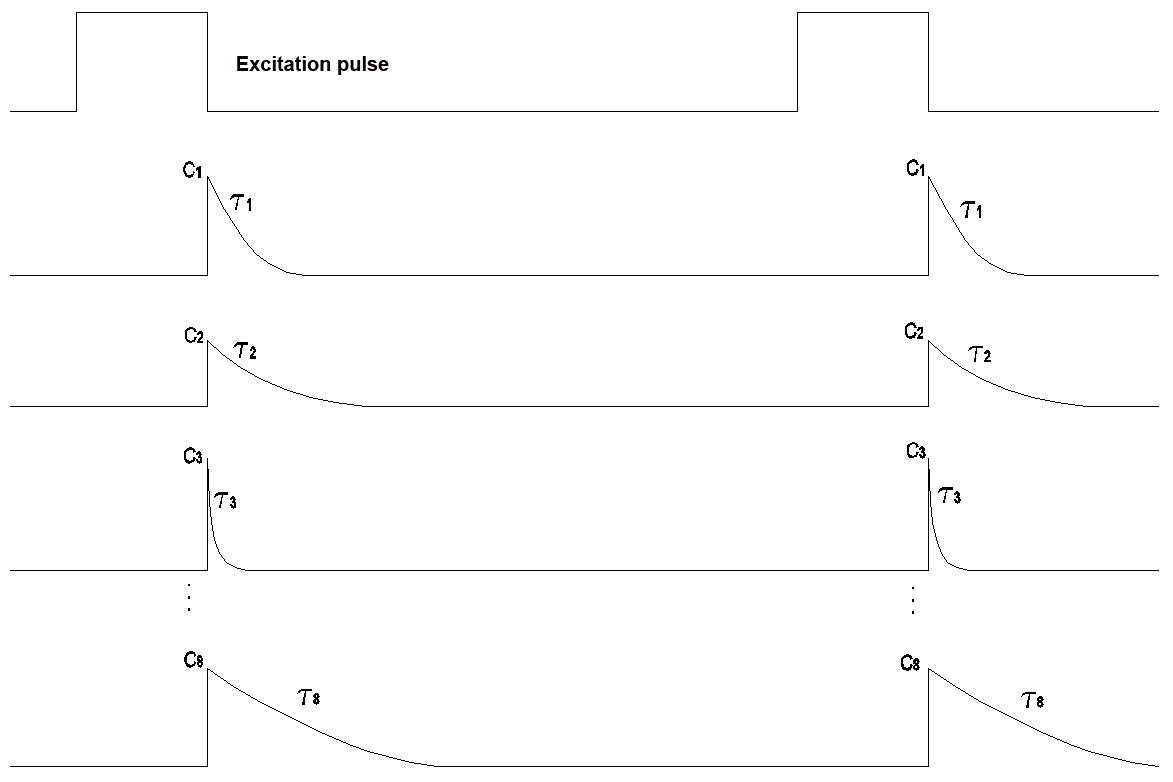
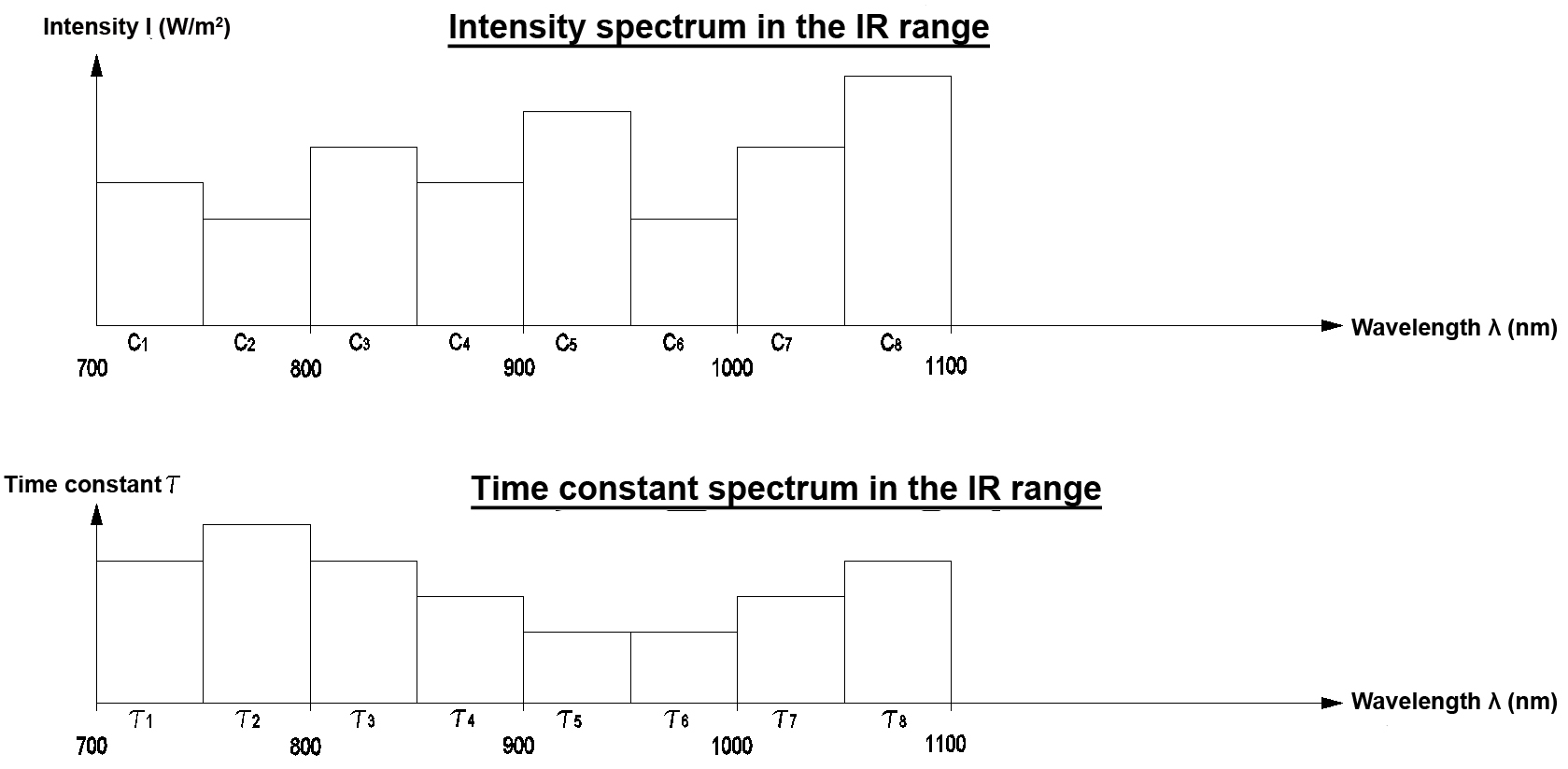
This version checks both the secondary spectrum and the relaxation behaviour in the IR range. The IR range is divided into 8 segments for this purpose. Both the time constant and the intensity of the secondary radiation are determined for every segment (patent pending for the process).

Corresponding software:
Windows® software LUMI-LAB-Scope
Measurement is performed with 8 detectors in the wavelength range from 700 nm to 1100 nm. Every detector checks a measurement window of 50 nm and determines the initial intensity C1 and the time constant t of the respective relaxation pulse.
Corresponding software:
Windows® software LUMI-LAB-Scope
They evaluate both the intensity and the time constant in the respective wavelength range.